
Such quantum chemical topology (QCT) methods include the electron localization function (ELF) and the quantum theory of atoms in molecules (AIM or QTAIM). The insights of VSEPR theory are derived from topological analysis of the electron density of molecules. Gillespie has emphasized that the electron-electron repulsion due to the Pauli exclusion principle is more important in determining molecular geometry than the electrostatic repulsion. Therefore, the VSEPR-predicted molecular geometry of a molecule is the one that has as little of this repulsion as possible. The greater the repulsion, the higher in energy (less stable) the molecule is. The premise of VSEPR is that the valence electron pairs surrounding an atom tend to repel each other. It is also named the Gillespie-Nyholm theory after its two main developers, Ronald Gillespie and Ronald Nyholm. Valence shell electron pair repulsion ( VSEPR) theory ( / ˈ v ɛ s p ər, v ə ˈ s ɛ p ər/ VESP-ər, : 410 və- SEP-ər ) is a model used in chemistry to predict the geometry of individual molecules from the number of electron pairs surrounding their central atoms. Shows location of unpaired electrons, bonded atoms, and bond angles. For more information on how electron configurations and the periodic table are linked, visit the Connecting Electrons to the Periodic Table module.Model for predicting molecular geometry Example of bent electron arrangement (water molecule). 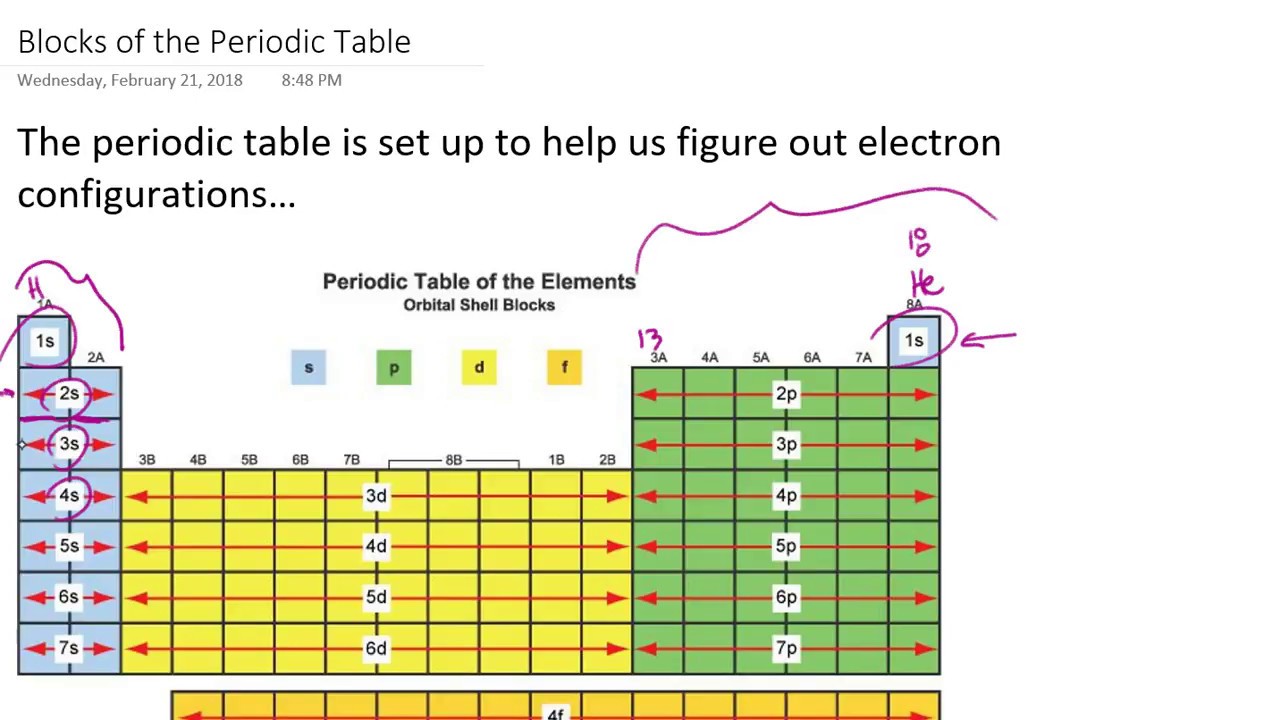
The periodic table is an incredibly helpful tool in writing electron configurations. Using the periodic table to determine the electron configurations of atoms is key, but also keep in mind that there are certain rules to follow when assigning electrons to different orbitals. The s-block is the region of the alkali metals including helium (Groups 1 & 2), the d-block are the transition metals (Groups 3 to 12), the p-block are the main group elements from Groups 13 to 18, and the f-block are the lanthanides and actinides series. Each orbital can be represented by specific blocks on the periodic table. The 1s orbital and 2s orbital both have the characteristics of an s orbital (radial nodes, spherical volume probabilities, can only hold two electrons, etc.) but, as they are found in different energy levels, they occupy different spaces around the nucleus. 
Orbitals on different energy levels are similar to each other, but they occupy different areas in space. The energy level is determined by the period and the number of electrons is given by the atomic number of the element. The p, d, and f orbitals have different sublevels, thus can hold more electrons.Īs stated, the electron configuration of each element is unique to its position on the periodic table. The four different types of orbitals (s,p,d, and f) have different shapes, and one orbital can hold a maximum of two electrons. Electrons exhibit a negative charge and are found around the nucleus of the atom in electron orbitals, defined as the volume of space in which the electron can be found within 95% probability. Every element on the Periodic Table consists of atoms, which are composed of protons, neutrons, and electrons. \)īefore assigning the electrons of an atom into orbitals, one must become familiar with the basic concepts of electron configurations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
